- Methylnaltrexone is significantly superior to placebo for relief of opioid-induced constipation (OIC).
- The use of methylnaltrexone to treat OIC was found to decrease the use of healthcare resources, increase work productivity, and reduce related system-wide healthcare costs.
- Both the 0.15 mg/kg and 0.30 mg/kg doses every other day, and the 12mg/day dose were found to improve the rate of rescue-medication free bowel movement (RFBM) within 4 hours of administration versus placebo.
- Systematic review and meta-analysis of randomized controlled trials included more than 1200 patients.
A meta-analysis and systematic review of randomized controlled trials has found methylnaltrexone to be significantly superior to placebo for the treatment of opioid-induced constipation (OIC). (Figure 1) The results were published recently in the Postgraduate Medical Journal.
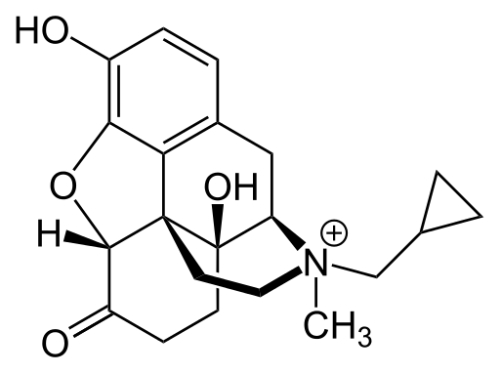
Severe constipation is a common adverse effect in patients receiving long-term opioid analgesia. Methylnaltrexone (Figure 2), a quaternary peripheral mu-opioid receptor antagonist, has been found to be highly effective for the treatment of OIC. Despite acting on the mu-opioid receptor, the agent appears to have no effect on opioid-related analgesia. Methylnaltrexone at 3 dosages was found to induce rescue-medication free bowel movement (RFBM) within 4 hours of administration.
The Analysis
The reviewers performed a systematic review and meta-analysis of randomized controlled trials using Cochrane Collaboration and MEDLINE data published as early as 2007. The study included all available literature related to methylnaltrexone, opioids, opioid receptors, opioid antagonists, and OIC in order to compare the effects of methylnaltrexone versus placebo. The primary observed outcome was RFBM within 4 hours of administration.
The analysis included 1239 patients of whom 599 received methylnaltrexone and 640 received placebo. The rates RFBM, ranging from 0.267 to 0.385, represented a statistically significant difference between the active agent and placebo (P < 0.0001). All 3 dosages tested—0.15mg/kg every other day, 0.30mg/kg every other day, and 12mg/day—were found to provide significantly superior relief of OIC compared to placebo.
Previous Data
A study published in Pain Practice in 2015 found similar results. This study examined the efficacy of subcutaneous methylnaltrexone for the relief of OIC in patients with advanced disease. The authors pooled data from 2 randomized, double-blind, placebo-controlled, phase 3 trials that examined the effects of 2 doses of subcutaneous methylnaltrexone (0.15mg/kg and 0.30mg/kg).
The authors reported that >50% of patients treated with methylnaltrexone (n=165) at either of the 2 doses experienced a RFBM within 4 hours of administration. Just 14.6% of the 123 placebo-treated patients reported RFBM within 4 hours of administration (P<0.0001 for both methylnaltrexone doses vs. placebo). The clinical response to treatment with methylnaltrexone was significantly greater than placebo response in all patient subgroups (P<0.01).
“The largest differences vs. placebo were observed for patients taking methylnaltrexone 0.30 mg/kg with a non-cancer primary diagnosis (70.0% [methylnaltrexone] vs. 12.8% [placebo]; P<0.001) and for patients taking methylnaltrexone 0.30 mg/kg maintained on ≥150 mg/day baseline morphine equivalent doses (73.3% vs. 16.7%; P < 0.0001),” the authors noted. The most common adverse events were abdominal pain, flatulence, and nausea.
Conclusions
The authors of both studies concluded that these results support the use of methylnaltrexone for the treatment of OIC. Given its effectiveness in the treatment of OIC, the use of methylnaltrexone appears to improve the efficiency of the use of healthcare resources, to improve work productivity, and to decrease system-wide healthcare costs.
