FDA Clears System for Monitoring Blood Glucose, Recommending Insulin Dosage
By Andrew John, /alert Contributor
May 19, 2021
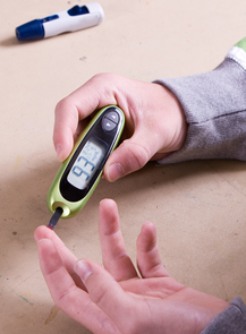
The FDA has given 510(k) clearance to the novel Bigfoot Unity Diabetes Management System, which monitors blood glucose and recommends personalized insulin dosage for patients with diabetes who use multiple daily injection therapy, Bigfoot Biomedical announced.
“Diabetes management is incredibly hard because insulin has no fixed dose or timing, leaving individuals to constantly determine their doses and configure devices as they make multiple critical decisions every day about how much insulin to take,” Jeffrey Brewer, CEO of Bigfoot Biomedical, said in a press release. “At Bigfoot, we want to ease the burden of diabetes for people taking insulin by minimizing the anxious guesswork involved with insulin dosing in a convenient, simple way.”

The device uses a mobile app, a Freestyle Libre 2 (Abbott) continuous glucose monitoring device and “smart” pen caps that synchronize with each other to measure the user’s blood glucose and display the patient’s recommended insulin dose. Users can view their current glucose values, track glucose trends over time and view their recommended insulin dosage based on a physician’s input using the monitoring device and the app, while the recommended dosage can also be read directly from a display on the smart pen caps. The system automatically logs user information including the time of dose and data on glucose levels and dosage amounts. This data is saved to a cloud where patients and care providers can view the log.
“For health care providers the Bigfoot Unity diabetes management program could be a gamechanger by connecting them to data that can provide better insight into what their patients are actually doing,” Jim Malone, MD, chief medical officer for Bigfoot Biomedical, said in the announcement. “By replacing guesswork and complexity with connectivity and simplicity, the Bigfoot Unity program is truly serving as that real-time partner for both patients and providers when it comes to dose decision support and overall diabetes management.”
Bigfoot also offers reminders and notifications, such as an automatic notification to tell users they have forgotten a long-acting insulin dose and an alert that tells users when their glucose values fall to less than 55 mg/dL. The system is appropriate for an array of patients, “from those with type 1 diabetes not on insulin pumps to those with type 2 diabetes needing to step up their insulin therapy,” the company said.
“There are more than 7 million people in the U.S. on insulin therapy and many use insulin injections multiple times a day, yet there have been few advancements for this hugely underserved market,” Brewer said in the press release. “Bigfoot Unity is specifically designed to be simple and accessible – no matter the person’s level of technical expertise – removing a key barrier to health equity. We know people with diabetes want to be successful with their insulin therapy, and it’s often complex and expensive technology that gets in the way.”
Disclosures: Brewer and Malone are both employees of Bigfoot Biomedical.








.jpg)