- Results of largest study to date were published in The New England Journal of Medicine.
- Randomized trial involved more than 9000 patients who had stable or unstable coronary artery disease.
- The primary outcome was a composite of death from any cause and nonfatal spontaneous myocardial infarction at 5 years of follow-up.
- Secondary outcomes included repeat revascularization, stent thrombosis, and quality of life.
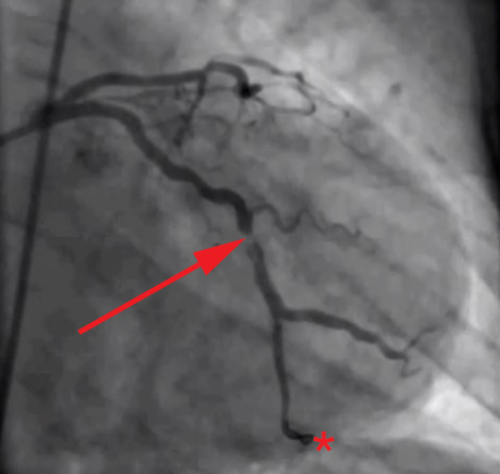
being treated with primary percutaneous coronary intervention, showing partial occlusion
of left circumflex coronary artery. The guide wire has traversed
the occlusion and its tip is visible at the bottom of the image.
(Sources: Wikipedia/By Todt T, Maret E, Alfredsson J, et al./Creative Commons.)
The largest study to date has found that the use of drug eluting stents does not reduce rates of all-cause mortality or death as a result of myocardial infarction (MI) (Figure 1). The stents do reduce rates of repeat revascularization when compared with bare-metal stents. These were the results of a large, randomized, multicenter trial published recently in The New England Journal of Medicine.
In patients undergoing percutaneous coronary intervention (PCI), there were no significant differences between those receiving drug-eluting stents (Figure 2) and those receiving bare-metal stents (Figure 3) in the composite outcome of death from any cause and nonfatal spontaneous myocardial infarction. Rates of repeat revascularization were lower in the group receiving drug-eluting stents. (The trial was funded by the Norwegian Research Council and others; NORSTENT ClinicalTrials.gov number, NCT00811772.)

(Sources: Wikimedia Commons/By Jack McLure/Creative Commons.)
The primary outcome was a composite of death from any cause and nonfatal spontaneous myocardial infarction after a median of 5 years of follow-up. Secondary outcomes included repeat revascularization, stent thrombosis, and quality of life 5 years after stenting.
The trial included 9013 patients who had stable or unstable coronary artery disease who due to undergo percutaneous coronary intervention (PCI). The patients were randomly assigned to undergo implantation of either a drug-eluting stent or a bare-metal stent. Everolimus- or zotarolimus-eluting stents were implanted in 96% of patients who received a drug-eluting stent.

(Sources Wikimedia Commons/By Mick Hensman/Creative Commons.)
Results
The researchers reported results at 6 years of follow-up. The rates of the primary outcome were 16.6% in the group receiving drug-eluting stents and 17.1% in the group receiving bare-metal stents. There were no significant between-group differences in the components of the primary outcome. The difference was not statistically significant (hazard ratio, 0.98; 95% confidence interval [CI], 0.88 to 1.09; P=0.66).
The results for repeat revascularization were different at 6 years of follow-up. Rates of any repeat revascularization were 16.5% in the group receiving drug-eluting stents and 19.8% in the group receiving bare-metal stents. These results were statistically significant (hazard ratio, 0.76; 95% CI, 0.69 to 0.85; P<0.001). Also statistically significantly different were the rates of definite stent thrombosis at 0.8% in patients receiving drug-eluting stents and 1.2% in patients receiving bare metal stents (P=0.0498). Quality-of-life measures did not differ significantly between the two groups.
The study found no significant difference between drug-eluting or bare-metal stents in either total patient deaths, nonfatal heart attacks, angina or even patient quality of life.
"The evidence in favor of contemporary drug-eluting stents over bare-metal stents may not be as strong as has been thought," said study author Kaare Harald Bønaa, MD, PhD, Professor of Medicine at the Norwegian University of Science and Technology in Trondheim, Norway. "Thirty patients would need to be treated with drug-eluting stents rather than with bare-metal stents to prevent one repeat revascularization," he noted.
