- Empagliflozin reduced the risk of cardiovascular death by 38%.
- Cardiovascular disease is the leading cause of death among diabetic patients.
- Reduced composite risk of MI or stroke and cardiovascular death by 14%.
- Study examined effects of empagliflozin 10mg or 25mg vs. placebo in more than 7000 diabetic patients.
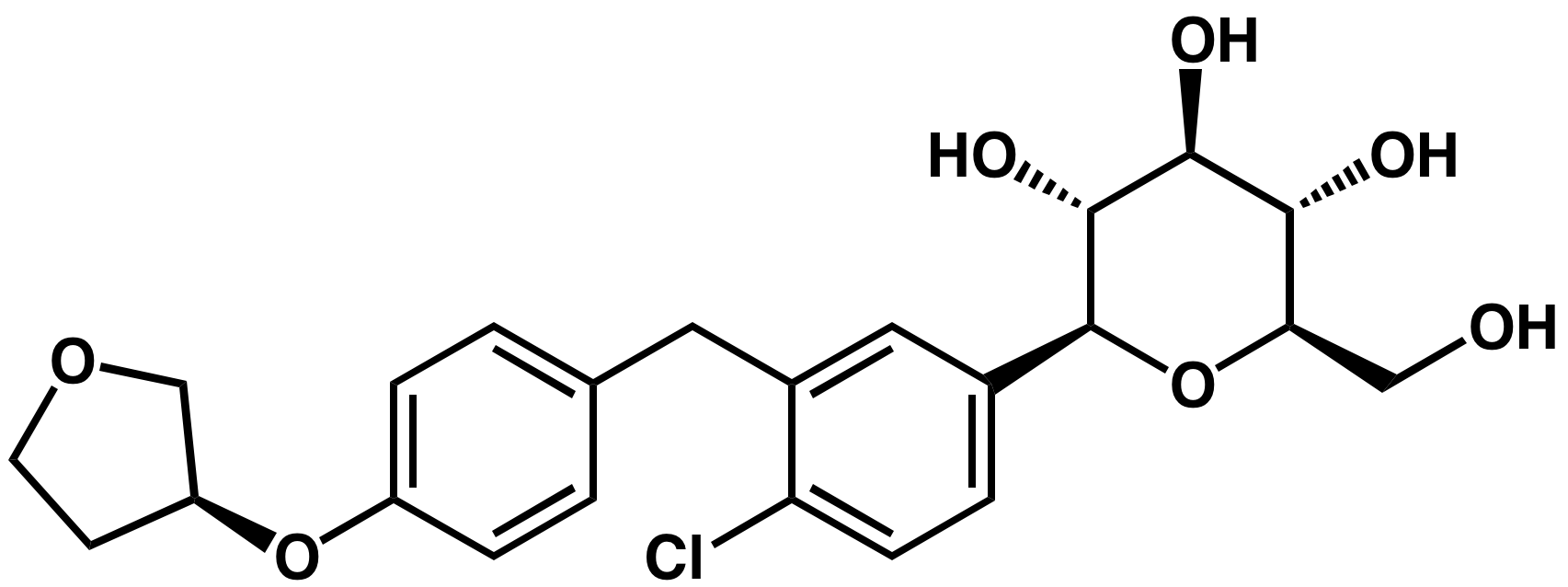
The diabetes drug empagliflozin (Figure 1.) has been shown to significantly reduce the risk of deadly heart disease in diabetic patients, a first for any such drug. The study was published in the New England Journal of Medicine.
Cardiovascular Disease
In a prospective, randomized, placebo-controlled clinical trial (n=7020), empagliflozin (Jardiance), sold jointly by Eli Lilly and Boehringer Ingelheim, reduced the overall composite risk of heart attack or stroke, or of dying from cardiovascular causes, by 14%. When the researchers isolated cardiovascular-related deaths, the reduction was 38%.
“It's almost like, how much more benefit can you get?” Silvio E. Inzucchi, MD, asked rhetorically in an interview at the 2015 annual meeting of the European Association for the Study of Diabetes where the results were presented. “Personally, I was shocked by the findings,” he noted. Dr. Inzucchi is Professor of Medicine (Endocrinology); Clinical Director, Section of Endocrinology; Director, Yale Diabetes Center; Director, Endocrinology & Metabolism Fellowship; Director in the Yale Affiliated Hospitals Program. He was part of the multicenter research team.

Silvio E. Inzucchi, MD
“Because cardiovascular disease [Figure 2.) is the leading cause of death for people with diabetes,” Dr. Nissen added, reducing that rate “by 38% is a landmark result.”
The Study
In the multinational, multicenter study randomly assigned 7020 patients to receive 10 mg or 25 mg of empagliflozin or placebo once daily. The primary composite outcome was death from cardiovascular causes, nonfatal myocardial infarction, or nonfatal stroke, as analyzed in the pooled empagliflozin group versus the placebo group. The key secondary composite outcome was the primary outcome plus hospitalization for unstable angina.
There were no significant between-group differences in the rates of myocardial infarction or stroke, but in the empagliflozin group there were significantly lower rates of death from cardiovascular causes (3.7%, vs. 5.9% in the placebo group; 38% relative risk reduction), hospitalization for heart failure (2.7% and 4.1%, respectively; 35% relative risk reduction), and death from any cause (5.7% and 8.3%, respectively; 32% relative risk reduction). There was no significant between-group difference in the key secondary outcome (P=0.08 for superiority).
Empagliflozin is a member of the gliflozin class of drugs which inhibit renal glucose reabsorption and reduce blood glucose (Figure 3).
“Patients with type 2 diabetes at high risk for cardiovascular events who received empagliflozin, as compared with placebo, had a lower rate of the primary composite cardiovascular outcome and of death from any cause when the study drug was added to standard care,” the researchers reported. The study was jointly funded by Eli Lilly and Boehringer Ingelheim.
